How to save your dog’s life with patent analysis
Visualization of Original Ugly Sister’s Patent Analysis

Dogs are our friends and family members. As dogs get older, many dogs will develop tumors like people. At present, domestic pet hospitals can provide very few treatment schemes for pet tumors, only surgery and chemotherapy, and radiotherapy for small animals is still prohibited in our country.
In recent years, there have been many breakthroughs in the treatment of human tumors, such as PD-1, CAR-T cell immunotherapy, etc. These methods have shown good therapeutic effects on tumors, and some can even be completely cured.
Therefore, when the dog who has been with us for more than ten years is old and has a tumor, and it can’t be operated and the chemotherapy effect is not good, of course we can’t just give up. Try to find out whether there is a tumor immunotherapy program suitable for dogs, inhibit tumors, let them live with cancer, and be happy and healthy to accompany us for a few more years.
Today, this article introduces the current scheme that can be used for pet tumor treatment by analyzing related patents.
PART 1 retrieval strategy
Technical scope of patent search: This search is mainly aimed at the technology of tumor immunotherapy involving pets. Therefore, the key words include: dog, cat, pet, cancer and tumor, and the IPC classification numbers involved are C12N (bio-related) and A61K or A61P (drug-related).
Geographical scope of patent search: the current search is to provide domestic pet owners with a treatment plan in China, and the technical feasibility in places too far away like the United States and Europe is not high. Therefore, the applicant’s address is limited to Chinese mainland and Japan (considering that Japan’s medical technology is developed and close to us, many Chinese patients go to Japan for treatment of cancer).
Manual reading and screening: after the number of patent documents is limited to the range that can be read manually through the above retrieval, manual reading is started to screen useful information. The basic requirement of the standard of artificial screening is to make it clear that it can be used for tumor treatment in dogs, and more preferably, it can give experimental data in the examples to confirm that it does have anti-tumor effect in dogs.
PART 2 analysis results
Through analysis, the treatment schemes that can be tried in domestic tumor immunotherapy for pets are sorted out as follows:
1. Beijing Weijiexin Biotechnology Co., Ltd
CN110590959A, recombinant canine PD-1 fusion protein, its preparation method and application, filed on September 19, 2019.
This patent application provides a recombinant canine PD-1 fusion protein, and in its example, it shows that this treatment scheme has been used in pet cats and dogs. It should be the second half of 2019 in a pet hospital in Beijing for clinical trials.

Therapeutic effect: The overall objective remission rate is 20%-30%.

The inventor of this patent application, Dr. Luo Haoshu, is an associate professor and master/doctoral supervisor of the School of Biology, China Agricultural University.

2. Beijing Conoco Fu Biotechnology Co., Ltd.
WO2019149039A1, an anti-tumor protein composition and its application, filed on January 9, 2019.
The patent application relates to an anti-tumor method using cytokine fusion protein, which comprises an anti-tumor composition, including proteins IL12, GMCSF and IL2, and achieves the technical effect of good inhibition on tumors at all stages and slight adverse reactions.
This cytokine fusion protein is a broad-spectrum anti-tumor drug. The protein anti-tumor drug is directly injected into the tumor to adjust the immune environment inside the tumor and realize the inhibition of the tumor. Experiments on sarcoma, oral melanoma and breast cancer in dogs and cats have been done in the examples, and the data show that most tumors have been suppressed, even shrunk and subsided.
Further search found that Conoco Clothing Company and the inventor of this patent, Zhang Jinyu, also applied for two other patents and published a paper in The Lancet’s sub-journal EBioMedicine. Although these two patents and journals did not explicitly say that they were used to treat Wang Wang tumor, their technical contents were very relevant to this application:
WO2018184484A1, cytokine combination, with the earliest priority date of April 6, 2017, relates to a cytokine combination, which comprises at least three cytokines, and the cytokines are selected from the following groups: IL12, GMCSF, FLT3L, IL2, IL15 and IL21;;
WO2020024922A1, protein heterodimer and its use, the earliest priority date is July 30, 2018, and it relates to a protein heterodimer, which comprises a first polypeptide chain and a second polypeptide chain, wherein the first polypeptide chain comprises IL12a and a first factor fused with IL12a, and the second polypeptide chain comprises IL12b and a second factor fused with IL12b, and the first factor and the second factor are selected from IL2 and GMCSF.
The paper "In Situ Administration of Cytokine Combinations Induces Tumour Regression in Mice" also involves the treatment of tumors with IL-12+GMCSF+IL-2.
Through reading the above patents, Xiao Bian understands that they use the cytokine composition to treat tumors (WO2018184484A1), then use protein heterodimer (WO2020024922A1) to make these cytokines into a cytokine fusion protein, and then use this cytokine fusion protein to treat tumors (WO2019149039A1), and treat tumors in dogs.
The applicant should think that this technology is more important and has better effect, so all the applications are PCT patent applications. Because they have been applied in recent two years, they have not seen the published texts of other countries.
3. Japan Quanyao Industry Co., Ltd.
Japan Quanyao Industrial Co., Ltd. has two patent applications in pet tumor immunotherapy.
Patent application 1: cn110603056a, a method for inhibiting the infiltration of regulatory T cells based on CCR4 blockade and a method for treating tumor diseases in dogs, filed on April 26th, 2018.
Patent application 2: wo2016006241a1, anti-canine -PD-1 antibody or anti-canine-PD-L1 antibody. The earliest priority date is July 9, 2014. This patent has been granted in Europe, Japan and the United States.
Founded in 1946, Japan Quanyao Industry Co., Ltd. is a research and development enterprise engaged in animal medicines. Its current animal drugs include some conventional veterinary drugs, such as anti-inflammatory, analgesic, fever-reducing, anthelmintic and so on.
However, judging from the above two patents, Japanese Quanyao should be engaged in the research and development of animal antitumor drugs. I continue to check the inventors of these two patents. They are not researchers of Quanyao in Japan, but researchers of Tokyo University and Yamaguchi University in Japan. This shows that Quanyao Industrial Co., Ltd. is cooperating with Japanese universities to develop animal antitumor drugs.
Continuing to search with the inventors of the above two patent applications, I found two Japanese scientific research institutions specializing in the treatment and research of Wang Wang tumor.
4. Laboratory of Veterinary Clinical Pathology, University of Tokyo, Japan
CN110603056A, a method for inhibiting the infiltration of regulatory T cells based on CCR4 blockade and a method for treating tumor diseases in dogs, by former Tian Zhen Wu.
The patent application relates to a pharmaceutical composition for treating canine tumors, which contains a compound that blocks the combination of canine CCL17 and canine CCR4 as an active ingredient. The pharmaceutical composition is a broad-spectrum antineoplastic agent, and can be used for treating transitional epithelial carcinoma, prostate cancer, breast cancer, malignant melanoma, squamous cell carcinoma and lung adenocarcinoma.
Before treatment, it is necessary to analyze the Treg (Regulatory T Cell) infiltration analysis in the tumor tissue of dogs and confirm the CCR4 expression in the tumor infiltration Treg. (Xiao Bian’s understanding: First of all, we should do pathological examination to see if the tumor of the dog has Treg infiltration and CCR4 expression. If so, the treatment with the pharmaceutical composition of this application may be effective. Just like treatment with PD-1, tumor cells need to express PD-1. Xiaobian is not in the biological field, please pat it if it is wrong)

Then, in the example of this patent application, the therapeutic effect data of pet dogs are given, which proves that the anti-CCR4 antibody (Mojiamuzumab) in this patent application is an effective therapeutic drug with less side effects on various tumors in dogs.

The inventor of this patent application is former Tian Zhen Wu of the Veterinary Clinical Pathology Laboratory of the University of Tokyo.

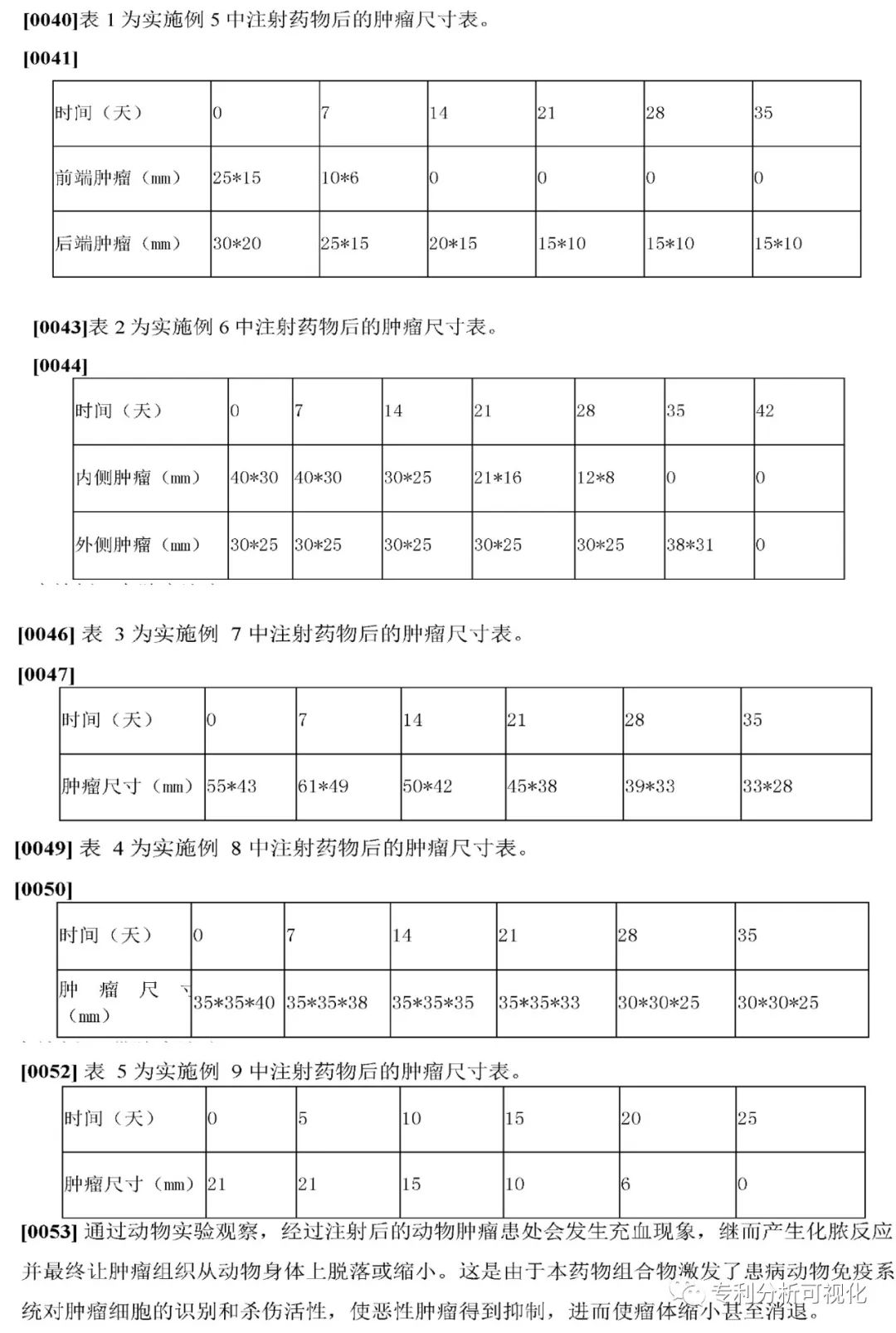
This Maeda teacher should be a very caring and enthusiastic person, and even found his personal website, which specifically introduces his veterinary clinical pathology laboratory at the University of Tokyo.

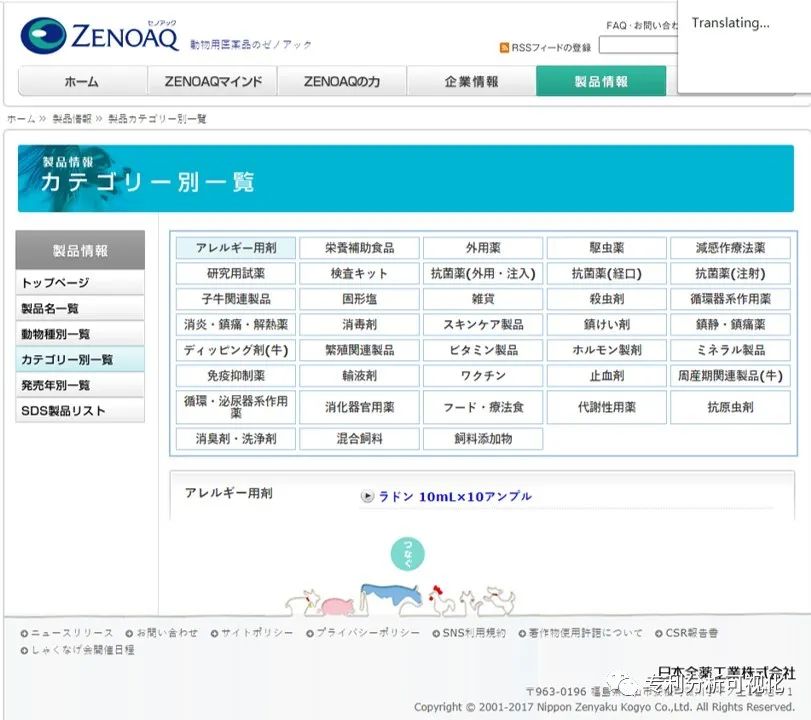
Moreover, their laboratory is currently recruiting clinical evaluation of anti-regulatory T cells (CLEAR), which is the anti-Treg treatment scheme related to the above patent.
The recruitment copy says:
がんをぅたちのためにののををぅぅさにのの12357
For Wang Wang who has cancer, for the shovel officer who thinks of his beloved Wang Wang, and for those who have cancer, it will be a great honor if this project can make a little contribution.
I hope this project can be successful and I can come to China in the future to give Wang Wang, who has cancer, more hope.
5. Laboratory of Veterinary Molecular Diagnosis and Therapy, Faculty of Common Veterinary Medicine, Yamaguchi University, Japan
WO2016006241A1, anti-canine -PD-1 antibody or anti-canine-PD-L1 antibody, inventor: Nobuya Mizuno.
This patent has been authorized in Europe, Japan and the United States. His inventor, Takuya Mizuno, is also a professor in the Laboratory of Veterinary Molecular Diagnosis and Therapy, Faculty of Common Veterinary Medicine, Yamaguchi University, Japan.

The research currently being carried out in the Laboratory of Veterinary Molecular Diagnostics and Therapeutics, where Professor Mizuno Toyo is located, includes:

Wang Wang oncolytic virus therapy, a kind of therapy that specifically infects cancer and destroys cancer cells. It has been proved that it can kill cancer cells in dogs to fight against various cancers.
Wang Wang PD-1 antibody drugs, including therapeutic methods for immunomodulation purposes, such as molecular suppression of immune checkpoints, and cytotoxic antibodies using antibodies against cancer cell surface molecules. This technology should be the PD-1 technology that was searched above and developed in cooperation with Japan Quanyao.
Wang Wang CAR-T immune cell therapy, which uses immune cells collected from dogs with cancer to prepare genetically modified immune cell CAR-T cells in vitro, is a very popular research direction of tumor treatment at present.
6. Beijing Baiao Saitu Gene Biotechnology Co., Ltd.
CN109452229A, preparation method and application of canine PD-1 genetically modified animal model, filed on November 19th, 2018.
The patent application provides an sgRNA capable of specifically targeting Pd-1 gene, a canine-derived animal model of PD-1 gene, a method for preparing an sgRNA vector of the canine-derived animal model, a preparation method of the canine-derived animal model and related applications, and a genetically modified canine-derived mouse is prepared, which can normally express PD-1 protein containing the functional domain of the canine PD-1 protein. The example of this patent provides relevant experimental data on tumor inhibition in mice, but there is no relevant patent on clinical experimental data of tumor in dogs.
Beijing Baiao Saitu Gene Biotechnology Co., Ltd. is a gene biotechnology company located in Daxing, Beijing, and its management team is all doctors in the field of biomedicine. At present, 84 patents have been applied, most of which are related to humanized genetic modification technology. This company mainly develops human antibody drugs, and only the above patent involves PD-1 in dogs.
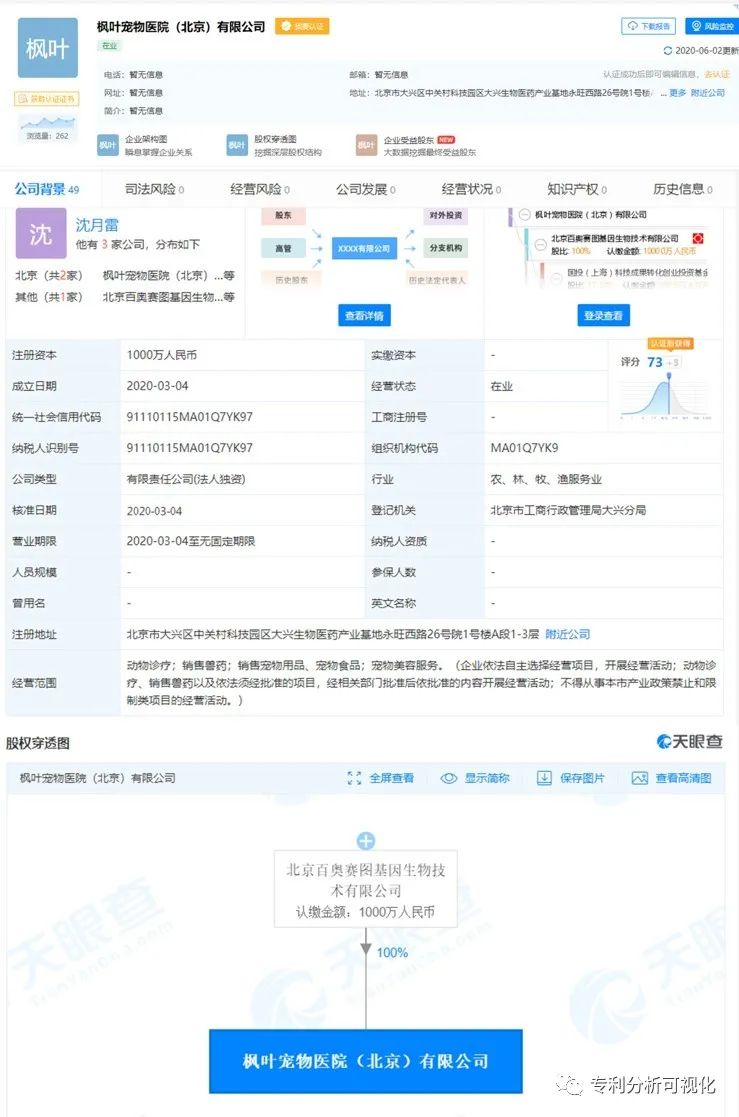
It is also found in the Eye of the Sky that Beijing Baiao Saitu has registered a Maple Leaf Pet Hospital (Beijing) Co., Ltd. in March this year, and it should start to enter the field of immunotherapy for pet tumors. But I haven’t seen them start business yet. I hope to get up and start business quickly and give the dogs more hope.
7. Beijing Xinuogu Biotechnology Co., Ltd.
CN110194799A, an anti-canine PD-1 antibody that binds to canine PD-1, filed on Jun. 3, 2019.
The patent relates to an anti-canine PD-1 antibody and a preparation method thereof, and the canine PD-1 antibody can be combined with canine PD-1 for application in drugs for treating canine cancer. The patent only discloses the experimental data of mice, but there is no clinical experimental data of dogs.
Xiao Bian has an impression on Sinovalley Biotechnology Company because they specialize in cloning cats and dogs, cloning 380,000 dogs.
Summary of analysis results of PART 3
At present, there are not many options to treat small animal tumors in China. After analysis, it is found that the following three are more reliable in terms of technical strength and experimental effect:
The cytokine fusion protein of Beijing Conoco Fu Biotechnology Co., Ltd. has been used in more than 300 cases of pets, most of which have achieved the effect of inhibiting tumor growth, and some of them have been completely relieved, and it is a broad-spectrum anti-tumor drug that can be tried;
Beijing Baiao Saitu Gene Biotechnology Co., Ltd., with strong technical strength, has 84 patent applications. It hopes to be used as a human medicine and also for the treatment of Wang Wang tumor. From the perspective of patent technology, it mainly involves PD-1 antibody, and their Maple Leaf Pet Hospital has just been established. It is expected that it will not start clinical trials for dogs until September this year, which is also a treatment plan worth looking forward to.
Japan Quanyao Industrial Co., Ltd., and its two scientific research institutions, the Laboratory of Veterinary Clinical Pathology of Tokyo University and the Laboratory of Veterinary Molecular Diagnosis and Therapeutics of the Faculty of Veterinary Medicine of Yamaguchi University, have entered the clinical trial stage. One is a regulatory T cell infiltration inhibition method based on CCR4 blockade, and the other is PD-1, which should be reliable, but it is not convenient to be applied in China at present, and it is recommended to Japanese shovel officials.
PART 4 Application of Patent Analysis Results
Ugly sister wanted to do this research because my ugly sister had been sneezing and running nose since the end of last year. At first, she thought it was rhinitis, but she didn’t get better after treatment for a long time. As a result, she went to the hospital for CT at the beginning of March this year and was highly suspected of being a nasal tumor.

An ugly younger sister with a runny nose when she sleeps.
The doctor said that if you have an operation, you should open your head, and it will recur soon. Ugly sister is 16 years old and can’t bear such a big operation. Chemotherapy is not effective for nasal cavity tumors. People who get nasal cavity tumors are usually treated with radiotherapy, but there is no place to treat small animals with radiotherapy in China at present.
Don’t you just let the tumor grow up? Now there are so many new methods for the treatment of human tumors, and our ugly sister can give it a try.
How can I know where I can give my dog tumor immunotherapy? So Ugly Sister analyzed all the patents about immunotherapy of dog tumors at present and found the above companies that are doing research in this field. After so many years of patent analysis, I finally feel useful.
Then, according to our actual situation, we can select the treatment options.
First of all, it is not convenient to go to Japan, so we ruled out two scientific research institutions in Japan first, but we are still looking forward to the related drugs of Japan Quanyao Industrial Co., Ltd. to achieve good results and successfully pass clinical trials.
Secondly, because the ugly sister is 16 years old, the ugly mother’s request is not to do any examination that requires general anesthesia, that is, not to do CT (dogs need general anesthesia when doing CT) and not to do pathological examination (because the tumor is in the nasal cavity, if you want to do pathological examination, you need to take samples with nasal endoscope under general anesthesia). Without biopsy, we don’t know what kind of tumor it is, and we don’t know if PD-1 is expressed.
So, in the end, we chose the cytokine fusion protein of Conoco. Because Conoco’s mature treatment plan is to inject drugs directly into the tumor, but our ugly sister’s nasal cavity tumor is in a bad position, so if she wants to inject it, she must be under general anesthesia, and ugly mother doesn’t agree. So we tried a new anti-tumor drug from Conoco Clothing Company, which was treated by intravenous injection, so that there was no need for anesthesia. So we ugly became the first Wang Wang in Chengdu to try this therapy.
On March 29th, Ugly Mommy took Ugly to the hospital to take venous blood, and the staff of Conoco Service Company took the blood sample away and sent it to their company’s laboratory. Then send it back after an operation in the laboratory.
On April 5th, Ugly received the first intravenous treatment.

Ugly receives the first treatment.
After treatment, I thought I would have a fever, but I didn’t have a fever at all.
I was going to have a second treatment the next week. As a result, just two days before preparing for the second treatment, Ugly suddenly had stomach cramps, pulled blood and vomited. The doctor diagnosed it as upper gastrointestinal bleeding, and quickly treated it. It took several days to stop bleeding and stop vomiting.
Because Ugly Ugly had digestive tract problems ten days before treatment, we don’t know whether this sudden upper gastrointestinal bleeding was caused by the poor digestive tract, the side effects of treatment, or the presence of lesions in the digestive tract.
However, Ugly Mom was afraid that Ugly would bleed if she was treated again, so she didn’t want to give Ugly a second treatment. We also want to get it in shape first.
Then Ugly Mom atomizes Ugly three times a day, and then drops some eye drops to stimulate it, so that it can sneeze hard to beat out the secretions inside.

Zhong Ugly does atomization three times a day.
On May 15, more than a month after the treatment, one day Ugly sneezed and sneezed a lot of secretions.
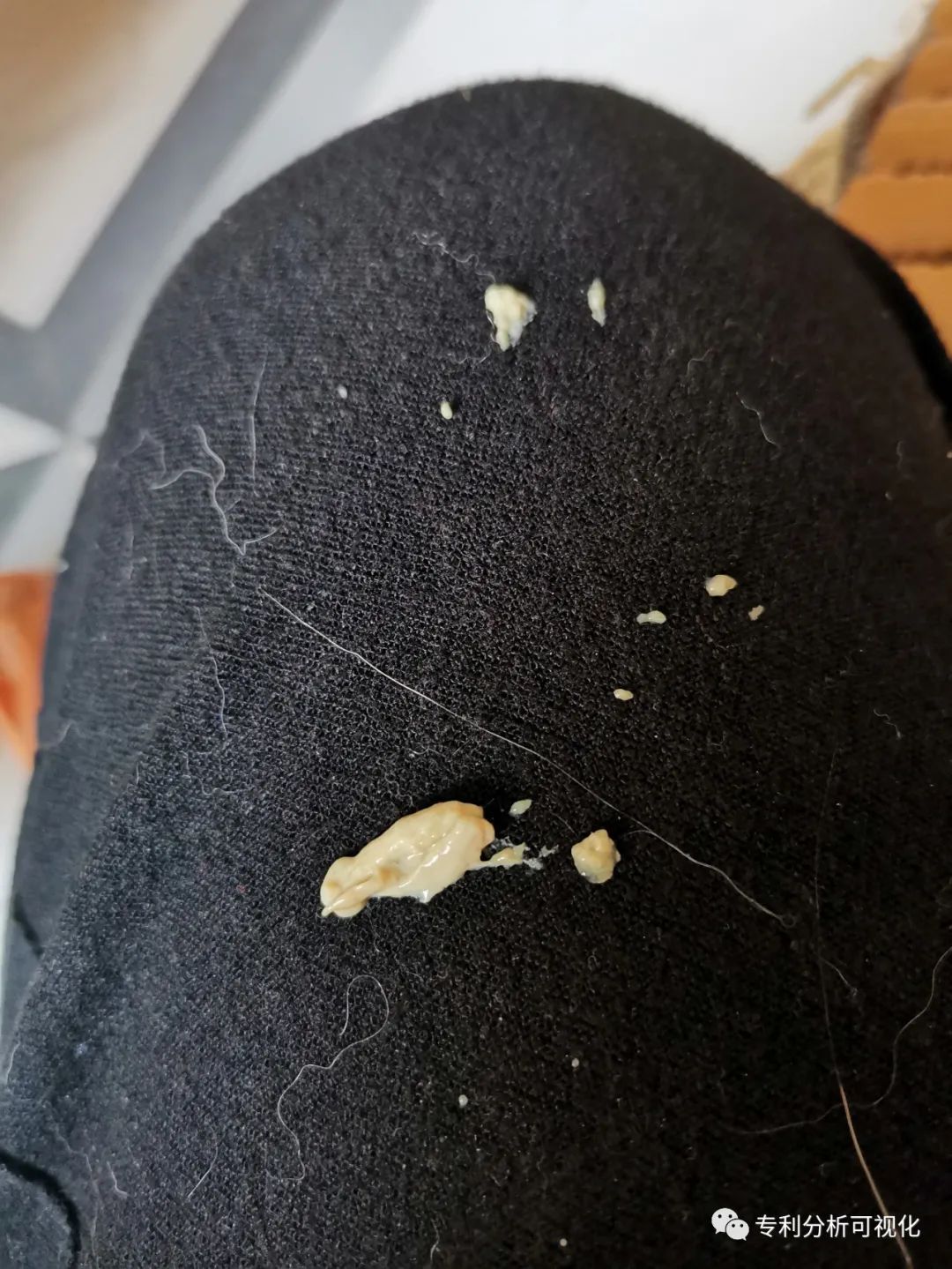
Ugly sneezing secretions from the nose.
Ugly mom said that the whole dog looks happier since it hit such a big lump, and it has to grind its teeth, which means that its nose doesn’t hurt.
On May 21st, I sneezed again and produced a lot of secretions.

Ugly sneezing secretions from the nose.
Then ugly mom said that ugly’s left nostril was open, and two points of water vapor could be seen when atomizing. In the past, the left nostril was blocked because of a tumor.

When atomizing, you can see the water vapor produced by the exhalation of two nostrils.
Now Ugly Ugly has completely stopped sneezing and running nose, and the previous symptoms have completely disappeared.
The staff of Conoco Clothing told us that it was a course of treatment for three times, but Ugly Mom saw the effect after the first treatment, and she was afraid that she would have stomach bleeding again, so she was hesitant to do it again.
Because ugly mom doesn’t want to take ugly to do CT, so we don’t know what the effect of tumor treatment is this time, just consider it effective. Ugly sister boldly guessed that the tumor might have shrunk, the nostril passage became larger, and all the secretions deposited in it were typed out, and then it was easy.
In a word, under the careful care of Ugly Mommy, the symptoms of Ugly Nose are gone now, and my mental state is good. Besides being clumsy when I am old, I can occasionally fight.

Ugly just celebrated her 17th birthday a few days ago. I hope she will always be healthy and happy.

postscript
Thanks for the technical support given by Lihua, Youhe and Teacher Mao, the small partners in the biological field, during the search for related patents; I would also like to thank Zhan Xing and Xin Rui for helping to contact the inventors. Thanks to the scientists and staff of Conoco Service, we can live a healthy life now.
Writing this article, I hope I can give you some information. If your master has a tumor, don’t give up. It would be a great honor if this article could make a little contribution.
↑ Long press to identify the QR code to follow ↑
Original title: How to Save Your Dog’s Life with Patent Analysis
Read the original text